Bovine embryo production and transfer biotechnology is an important asset to ensure speed of cattle farming genetic gain. In recent years, we’ve seen a global increase in cattle embryo production, reaching, in 2021, almost 1.5 million bovine embryos transferred according to the data collated by the International Embryo Technology Society (IETS).
The process of embryo production and subsequent transfer, rely on two types of techniques:
- in vivo embryo production, based on the superovulation of the donor cow using FSH, which causes the development of multiple follicles that are induced to ovulate at the same time. The female is inseminated with the sperm of a valuable sire, and 7 days after the uterus is flushed to collect the developed embryos.
- in vitro embryo production using oocytes aspirated from the ovaries of a donor (heifer or cow), which get fertilised in vitro with the semen of the desired sire. Following this process, the presumptive zygotes are cultured in vitro and then after evaluation of their quality.
In both cases the viable embryos can be transferred into a recipient female or frozen for unlimited storage.
The in vivo technology has been developed for the first time in cattle in 1920 in the UK from Rowson and colleagues, but it took another 50 years until 1970 for this technology to really become a commercially valuable reality for the breeding dairy and beef industry. With commercialisation of porcine pituitary extracts FSH/LH superovulation was made possible. More recently, with the generation of recombinant bovine FSH these technologies are used as a tool to maximise the genetic output of a female bloodline.
The use of embryo production allows ultimately the maximisation of the usage of the genetic potential of donor cows, superovulation requires donors to be at least pubertal and the initiation of the process. The level of embryos averagely collected from cows and heifers has not changed over the years; it has remained quite stable, with an average collection between 6 and 8 transferrable embryos per flushing.
The flushing procedure can be performed only in non-pregnant animals that have passed at least 45 days from parturition. Meanwhile, they have recovered a functional cyclicity and have perfect uterine health. The superstimulatory procedure cannot be repeated before 45 to 60 days from the last flushing; these are limiting factors that are less relevant for the use of in vitro embryo production. In fact the application of OPU can be performed even in prepubertal heifers as well as pregnant cows during the first trimester, as this procedure aspirates oocytes contained inside the follicles that are detectable from an ultrasonografic examination.
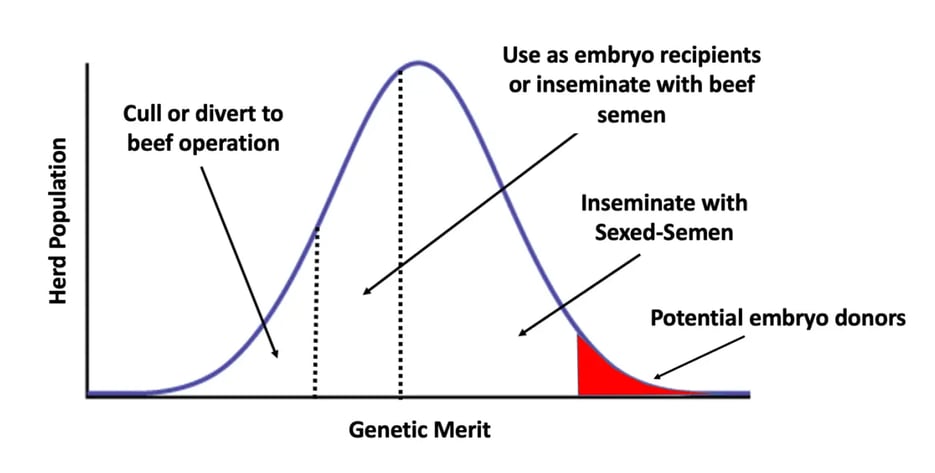
In the last decade the introduction of genomic testing at commercial level has allowed to identify 4 population of animals, and their characteristics can be correlated to a correct breeding program involving embryo transfer:
- the top 2.5%-5% of the herd which can be used as embryo donor either from superovulation or OPU,
- another 40-45% of the herd which can be selected to receive sexed semen, and their offspring maintained as replacements for the future,
- another 15% of animals which are good candidate to be used as embryo recipients or eventually to be inseminated with beef semen as their offspring will not improve the farm performances. Their lactations will still be an asset for the farm but their offspring output will either be from a higher genetic merit animal (embryo donor) or it will be diverted to the beef chain ensuring a higher return when sold as calves.
- The fourth population of cows with the poorest genetic merit, as their potential contribution to the future of the herd is predicted to be negative, should be considered for selling
The insertion of embryo transfer in the breeding strategy of a given dairy farm, allows to import genetic potential of females from any part of the globe to match at best the genetic characteristics of the herds with the unlimited combinations to improve the health and productive performances of their animals.
The use of embryo transfer for specific uses
Embryo transfer can be used in dairy cows to improve pregnancy establishment vs artificial insemination in 2 different conditions.
The first is during periods of heat stress. Most of the infertility caused by heat stress is because of damage to the oocyte and early embryo, these two are the most heat vulnerable elements of the breeding process. By the time an embryo is transferred, at the blastocyst stage, the embryo has acquired resistance to the maternal environment thermally stressed. As a result, there can be a large improvement in pregnancy rate using ET in summer, and there is little or no seasonal variation in P/ET even in hot climates. For this reason, using embryo transfer to substitute AI during the summer period is a way to stabilise pregnancy establishments per month in countries with hot summers.
The other situation in which ET can improve pregnancy outcomes is for the repeat-breeder cow. These cows are usually defined as those that have been inseminated 3 or more times without the establishment of pregnancy. Several studies have shown that pregnancy success after ET in repeat-breeder cows can be greater than for AI. One of the largest study, from Rodrigues et al., 2007, analysed the results from 5,693 AI and 3,858 ET in repeat breeder Holstein cows where pregnancy rates were greater after ET (41.7%) than after AI (17.9%).
The reason why ET improves fertility is unclear and causes of the repeat-breeder syndrome are likely to differ between individual cows and between herds of cows. Some causes of the repeat breeder syndrome, such as reduced oocyte quality could be improved by ET, the oocyte step is bypassed as the cow used as recipient will benefit from the oocyte of the donor animal, whereas other causes, such as alterations in uterine function, would not be impacted by ET.
Conclusions
Embryo transfer is a well-established technology for the breeding of cattle. The use of either in vivo or in vitro production consent the exploitation of the maternal lineage genetic potential at a much higher level than artificial insemination. Embryo transfer consent the infusion of genetics from any parts of the World without the needing of the transfer of live animals, avoiding the biosecurity hazards, consent the exploitation of low genetic potential females as producers of valuable offspring when used as recipients, and could improve breeding performances compared to AI in repeat breeders and in cows exposed to heat stress.
References
Betteridge KJ, Rieger D. Embryo transfer and related techniques in domestic animals, and their implications for human medicine. Hum Reprod. 1993 Jan;8(1):147-67. https://www.doi.org/10.1093/oxfordjournals.humrep.a137864.
Rodrigues, C. A., H. Ayres, R. M. Ferreira, A. A. Teixeira, R. F. Mancilha, M. E. F. Oliveira, A. H. Souza, and P. S. Baruselli. 2007b. Comparison of pregnancy rates after artificial insemination or embryo transfer in high producing repeat breeder Holstein cows. Acta Sci. Vet. 33(Suppl.1):1255 (abstr.).
About the author
Federico Randi (Ruminants Global Technical Manager)
Federico Randi is Global Technical Manager for Ruminants at Ceva Animal Health, specializing in cattle reproduction. With a Doctor of Veterinary Medicine degree “cum laude” from the University of Bologna, he focused his career on improving efficiency and sustainability of farmed animals. Randi conducts research on ruminants fertility, using technologies like Timed AI, embryo transfer, and recombinant technologies. His extensive experience includes collaborative projects with over 20 research institutions globally. He earned his PhD at University College Dublin, concentrating on fixed-time artificial insemination and embryonic maternal communication in cattle. Currently, he serves as a Board Member in the Scientific Commission of Animal Physiology for the European Federation of Animal Science (EAAP) as an Industry Representative.




Leave your comments here